 
The impressive piece here is long-term survival. Those weren’t directly compared but nivolumab and ipilimumab were superior to chemotherapy with a hazard ratio for survival of 0.77. What we saw here is that the clear winner was dual checkpoint blockade outperforming nivolumab alone and outperforming chemotherapy alone. Brahmer, MD, MSc, are the 5-year results. These data were presented at ASCO 2022 by Julie R. CheckMate 227 was a positive study, and this is an FDA-approved regimen we have long-term follow-up. Liu: That’s an interesting point, we should just be excluding the driver-positive lung cancers. That, I think, misled the plan of the trial around the primary end point and, in retrospect, I’m not surprised at all that it looks like the PD-L1–negative subgroup is the one that might benefit the most. Usually tumors are low for PD-L1, so I think we interpreted that that it was the PD-L1–positive patients who were benefiting. There were 12 patients with EGFR mutations, is a big number. Among the 77 patients who had a biomarker analysis for PD-L1, we looked at activity. Because it was a phase 1 trial, we included patients with actionable alterations. Neal Ready, MD, PhD: In the phase 1 trial we established that the full dose of nivolumab and ipilimumab 1 mg/kg was safe in patients with lung cancer. decision to focus on the PD-L1positive group. Dr Ready, you were involved in the early studies. The primary end point was observed in data from the PD-L1positive subgroup and that’s going to have important implications in how we interpret and maybe, more importantly, how the regimen was approved. 
The thought here was that giving nivolumab alone to PD-L1–negative disease was not well advised. If a patient had PD-L1 expression of 1% or greater they were randomly assigned to nivolumab and ipilimumab plus standard chemotherapy or nivolumab alone, whereas if they had PD-L1–negative disease 3 arms nivolumab/ipilimumab, chemotherapy alone, or a third arm that was nivolumab with chemotherapy. Patients with squamous and nonsquamous histologies were stratified and this split up into almost 2 different studies. It was for patients with advanced NSCLC who had no prior therapy and excluded EGFR and ALK mutations. CheckMate 227 was a bit of a complicated study. Liu, MD: The combination of nivolumab and ipilimumab has to be very effective in melanoma some early success was observed in lung cancer including small cell lung cancer. 1,2ĭuring the seminar, Ready and Liu discussed the new data and provided commentary on treatment considerations for patients who are eligible to receive these regimens. 1-4 Updated findings for both trials, 5- and 3-year data, respectively, were presented at the 2022 American Society of Clinical Oncology (ASCO) Annual Meeting ( Tables 1 and 2). Both play an important role in priming immune cells,” Liu continued.ĬheckMate 227 (NCT02477826) and CheckMate 9LA (NCT03215706) evaluated the immunotherapy combination of nivolumab (Opdivo), a PD-1 inhibitor, and ipilimumab (Yervoy), a CTLA-4 inhibitor, for patients with treatment-naïve NSCLC. “The other category we have is dual checkpoint blockade 2 different checkpoint inhibitors: one targeting PD-1 and the other targeting CTLA4. 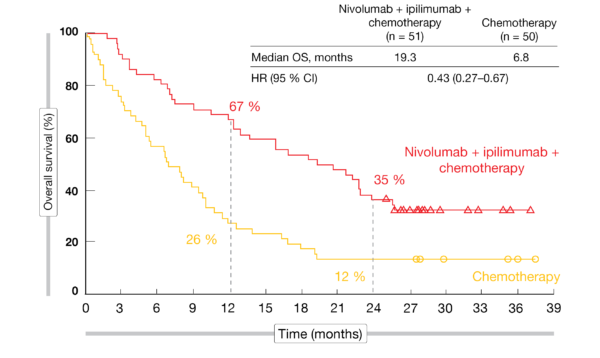
Liu, MD, who joined Ready in the seminar. “We have a lot of different options depending on histology and, presumably, more to come,” said Stephen V. In addition to monotherapy, chemoimmunotherapy treatment options have expanded the armamentarium for these patients. Immunotherapy treatment options for patients with NSCLC with PD-1 or PD-L1 inhibitors have demonstrated durable responses with longer-term follow-up. “For PD-L1–low expressing tumors, we have a certain set of possible treatments for PD-L1–high expressing tumors different options including monotherapy and other approaches.” “We can now identify the right treatment for the right patient I know that there’s a tendency to want to have one-size-fits-all treatment,” Ready said during a recent OncLive ® Seminar Series. At the time of diagnosis of advanced lung cancer molecular testing is imperative and the best marker to have data for is PD-L1, according to Neal E. Biomarker selection has played a crucial role in the rapid advancement of clinical benefit observed for patients with non–small cell lung cancer (NSCLC). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
